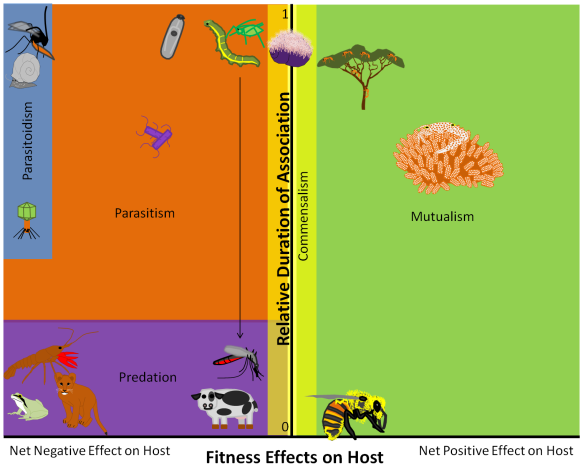
You can find ants and plants in almost every terrestrial habitat on the planet. Both groups can be incredibly abundant, so it isn’t surprising that ants and plants interact a lot. But some plants and ants have intimate symbiotic relationships that go far beyond the occasional interaction. Some of my favorite ecological stories involve these symbioses, and I’m going to post those stories in the coming weeks. But this week, I just want to introduce you to the system and let the insane photography skills of Alex Wild bring these organisms into your life.
Myrmecophytic plants: Who are they, and what do they provide their ant symbionts?
There are many genera of myrmecophytic plants, including flowers, shrubs, trees, and even ferns. These plants vary widely in the degree to which they invest in their ant symbionts. Below is a list of the structures that plants have evolved to provide their ants with resources, but not all ant-plants have all of these structures.
Domatia: Domatia are hollow structures that the ants can use for nests. Depending on the plant species, these domatia may be hollow stems or spines. For instance, check out this hollow thorn on an Acacia tree and the hollow base of this epiphytic plant. Both are homes for ants!



Extrafloral nectaries: Many plants provide nectar rewards in their flowers in order to attract pollinators. Ant-plants may also have extrafloral nectaries – structures that provide nectar but that are not associated with flowers. On ant-plants with domatia and active ant colonies, these extrafloral nectaries can feed resident ants. On ant-plants without domatia, these nectaries can attract ant visitors. Here are two gorgeous ants feeding at an extrafloral nectary.

Food Bodies: Ants can’t just survive on nectar; they need resources other than sugar, too. Some ant-plants have evolved to produce food bodies that contain proteins or lipids that ants can harvest for those vital nutrients. Here’s an ant harvesting one such food body.

Plant-ants: Who are they, and what do they provide their plants?
There are also many genera of ants that have symbiotic relationships with their plants. These ants can be facultative or obligate symbionts (meaning that they are only found living on plants), depending on the species. In the coming weeks, I’ll talk a lot more about the services that ants do (and do not) provide to plants, but here are the main points:
Defense: Plant-ants are feisty plant protectors! They can bite and sting herbivores or even throw the herbivores off the plant. Plant-ants will also attack competing plants. For instance, they will bite encroaching vines or inject neighboring plants with formic acid. (Yeah. For real. Look up Devil’s Garden.) Here are some ants getting rid of a vine, and some different ants hauling away an intruder ant.


Nutrients: When ants die and defecate, they fertilize their plants. (I bet you can imagine this one without a photo. Also, arboreal earthworms are a thing, and plants eat their poop, too. You’re welcome.)
Seed Dispersal: Some plants produce seeds with tasty exterior food bodies called elaiosomes. The ants collect these seeds and eat off the elaisomes, then put the seeds in their waste piles. During this process, the seeds are dispersed, and they’re also protected from predators while they’re in the ant nests.

Aren’t plant-ants and ant-plants cool?! You should check out Alex Wild’s website for more awesome photos. Stay tuned for more on plant-ant ecology next week!